Aim
The aim of this experiment was to investigate the use of anti-CD47 antibodies and examine its use as a targeted therapy for human solid tumours.
Results
CD47 is expressed on solid tumour cells
Weissman and his colleagues explored the expression of CD47 on a range of cancer types including: ovarian, breast, colon, bladder, glioblastoma, hepatocellular carcinoma and prostate. They used the technique of quantitative flow cytometry and immunofluorescence to detect expression.
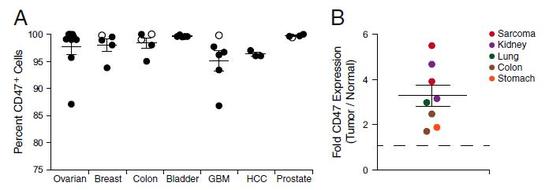
The results, as shown in Figure 2, indicate that CD47 is overexpressed on cancer cells by 3 fold when compared to normal expression levels.
Weissman and his colleagues explored the expression of CD47 on a range of cancer types including: ovarian, breast, colon, bladder, glioblastoma, hepatocellular carcinoma and prostate. They used the technique of quantitative flow cytometry and immunofluorescence to detect expression.
The results, as shown in Figure 2, indicate that CD47 is overexpressed on cancer cells by 3 fold when compared to normal expression levels.
CD47 mRNA expression levels predict survival
Gene expression data from previous cohorts of patients with gliomas, glioblastomas and ovarian cancer were investigated to determine whether CD47 mRNA expression levels can be used as a prognostic marker. The study utilised statistical analyses to stratify the patients based on high/low CD47 expression. The results indicated that patients with a high CD47 mRNA expression exhibited a decreased likelihood of progression-free and overall survival.
Anti-CD47 antibodies enable phagocytosis and inhibit tumour growth
A number of blocking and non-blocking antibodies (used as controls) were utilised against CD47 to determine the effects of blocking this surface molecule. Weissman and his colleagues found that the tumour cells treated with blocking anti-human CD47 mouse antibodies were effectively phagocytosed by both mouse and human tumour-associated macrophages (TAMs). In addition, the researchers also investigated the effect of the therapy on cancer stem cells and found that anti-CD47 antibodies were able to induce phagocytosis in these cell types as well.
The video below depicts a movie generated by the researchers in this study. It shows the phagocytosis of human tumour cells, following anti-CD47 antibody treatment. Notice the red macrophages 'eating' green tumour cells.
Gene expression data from previous cohorts of patients with gliomas, glioblastomas and ovarian cancer were investigated to determine whether CD47 mRNA expression levels can be used as a prognostic marker. The study utilised statistical analyses to stratify the patients based on high/low CD47 expression. The results indicated that patients with a high CD47 mRNA expression exhibited a decreased likelihood of progression-free and overall survival.
Anti-CD47 antibodies enable phagocytosis and inhibit tumour growth
A number of blocking and non-blocking antibodies (used as controls) were utilised against CD47 to determine the effects of blocking this surface molecule. Weissman and his colleagues found that the tumour cells treated with blocking anti-human CD47 mouse antibodies were effectively phagocytosed by both mouse and human tumour-associated macrophages (TAMs). In addition, the researchers also investigated the effect of the therapy on cancer stem cells and found that anti-CD47 antibodies were able to induce phagocytosis in these cell types as well.
The video below depicts a movie generated by the researchers in this study. It shows the phagocytosis of human tumour cells, following anti-CD47 antibody treatment. Notice the red macrophages 'eating' green tumour cells.
Anti-CD47 antibodies prevent tumour metastasis
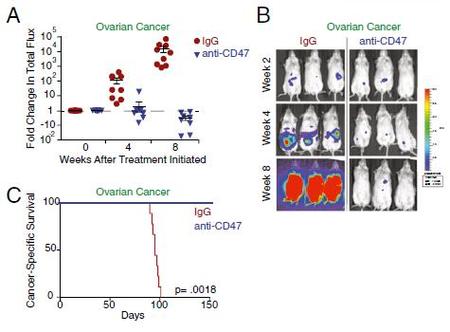
The study then focuses on the effect of the therapy on xenotransplantation models. Essentially, ovarian tumour cells from patients were injected into the peritoneal cavity of immunodeficient mice. It is important to note that a non-blocking control was not used for this section due to unavailability.
Tumour growth was monitored through bioluminescent imaging of GFP (previously transduced into the cells). The researchers discovered that the therapy had improved the survival of the mice and inhibited tumour growth as shown in Figure 3.
The study then focuses on the effect of the therapy on xenotransplantation models. Essentially, ovarian tumour cells from patients were injected into the peritoneal cavity of immunodeficient mice. It is important to note that a non-blocking control was not used for this section due to unavailability.
Tumour growth was monitored through bioluminescent imaging of GFP (previously transduced into the cells). The researchers discovered that the therapy had improved the survival of the mice and inhibited tumour growth as shown in Figure 3.
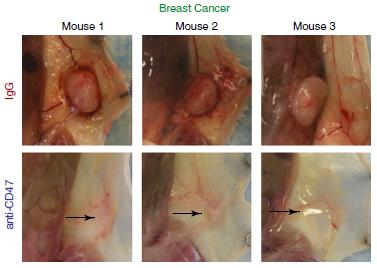
Breast cancer cells were also injected into mammary fat pads of the mice and anti-CD47 therapy was initated after the tumour was established. The results indicated that the tumours either completely regressed or growth was dramatically inhibited (Figure 4). However, one breast cancer sample did not change in tumour growth. These results also correlated to other xenotransplantation models for glioblastoma, colon cancer and bladder cancer.
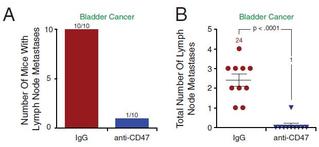
The researchers also examined the effect of the antibody therapy on metastasis. Through the use of different xenotransplantation models, they discovered that the therapy also decreased the incidence of metastasis, particularly in bladder cancer (Figure 5), lung cancer, and head and neck squamous cell carcinoma.
The researchers also examined the effect of the antibody therapy on metastasis. Through the use of different xenotransplantation models, they discovered that the therapy also decreased the incidence of metastasis, particularly in bladder cancer (Figure 5), lung cancer, and head and neck squamous cell carcinoma.
Anti-mouse CD47 antibodies inhibit tumour growth in immune competent host
The researchers finally tested the effectiveness of this therapy in immune competent hosts and found that the tumour size still regressed significantly in these models. In addition, the incidence of toxicity was also investigated and the researchers discovered there was minimal toxicity with isolated cases of short-term anaemia. This led to the conclusion that anti-CD47 therapy does not have a therapeutic benefit through direct toxicity.
The researchers finally tested the effectiveness of this therapy in immune competent hosts and found that the tumour size still regressed significantly in these models. In addition, the incidence of toxicity was also investigated and the researchers discovered there was minimal toxicity with isolated cases of short-term anaemia. This led to the conclusion that anti-CD47 therapy does not have a therapeutic benefit through direct toxicity.